Research
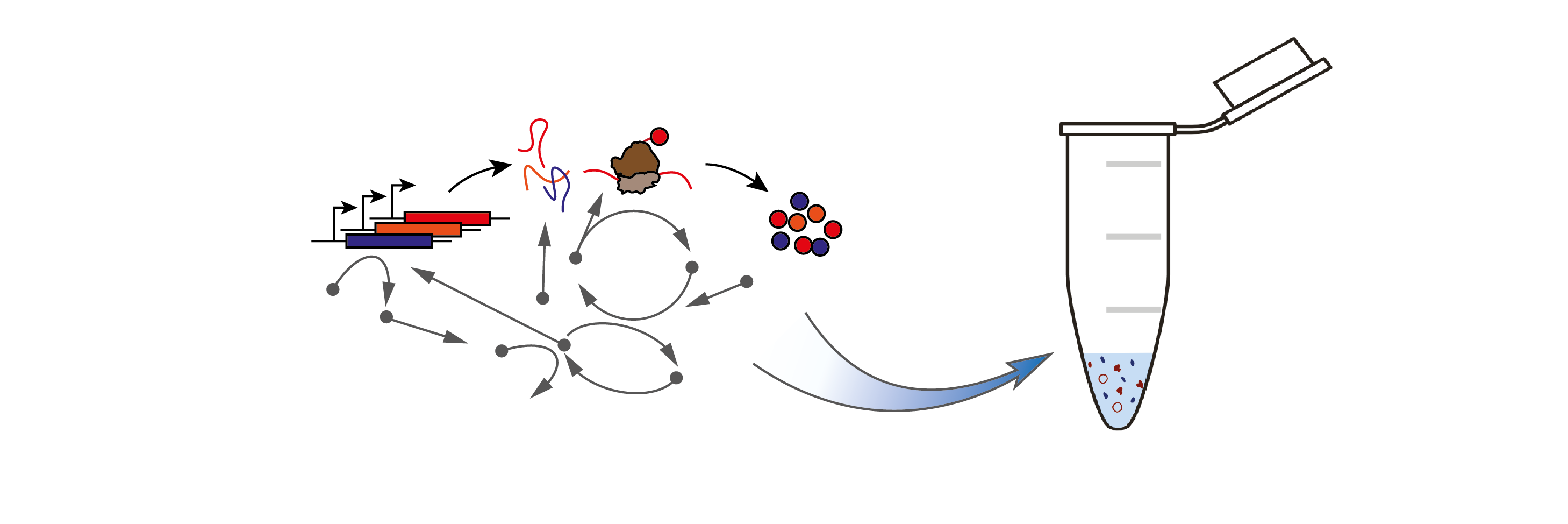
Synthetic biology is a rapidly growing field set to make profound impact in numerous industries including manufacturing, healthcare, agriculture, and sustainable energy, as well as our fundamental understanding of life itself. However, engineering synthetic biological systems reliably has proven to be a tremendously challenging task.
One promising approach is to use cell-free gene expression systems. These are in vitro systems that mimic the cellular environment and can be integrated with microfluidic technologies to rapidly prototype and screen synthetic gene circuits, prior to their deployment in vivo. This speeds up the synthetic biology design cycle, but also perhaps more powerfully, releases gene expression from the confines of the cell. In this way, the cell-free extracts themselves can be viewed as functional aqueous solutions, programmed by DNA.
The aim of our research is to harness these in vitro biomolecular mechanisms in a controlled and quantitative manner.
The consequence of this approach is twofold: the first is a deeper understanding of life, as we attempt to control and manipulate genetic and metabolic systems which have been crafted over billions of years of evolution. The second is the development of powerful cell-free applications which, unlike synthetic cellular systems, are not limited by the strict requirements to maintain life.
Funding
We gratefully acknowledge support from the following organizations:






